
Norah Hornsveld defends her PhD-thesis, titled Atomic layer deposition for Li-ion batteries: Novel chemistries, surface reactions and film properties on Wednesday 26th of January. Next to that, they can be used to apply a uniform thin layer to 3D structures," Hornsveld said. 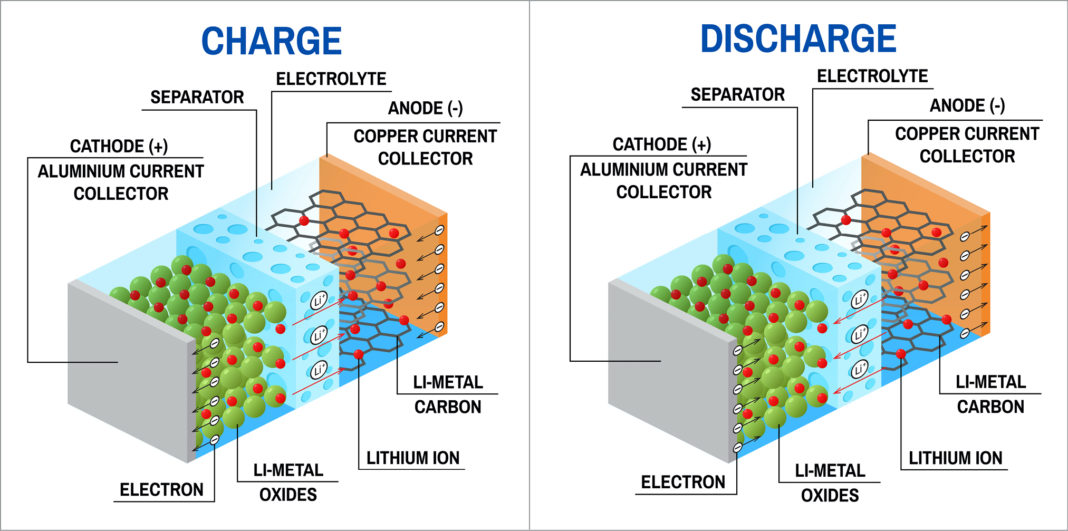
"My research suggests that plasma ALD processes could also be very interesting for making battery materials, because these offer more freedom in process parameters in order to adjust the properties of the layers. The solid-state battery is therefore safer and has a higher energy density. 
In this battery, the electrolyte is a solid material rather than a liquid, so there are fewer problems at the electrode-electrolyte interface. She also thinks her work will contribute to the development of next-generation batteries, such as the ‘solid state battery’. "We show that even a small change - ultra-thin in this case - can be a big improvement." Although her research is fundamental, she expects that certain processes can be applied on a large scale. With the developed processes, Hornsveld hopes to give more insight in the possibilities to improve the battery interface and provide a protective layer for the electrodes. This is important to be able to adapt the film to specific electrode materials." Solid-state battery "We were able to make films of different materials, with variable properties such as composition, layer thickness and crystallinity. “I looked at how you can influence the properties of the ultra-thin films by changing the ALD process parameters.” This is particularly interesting for applying a uniform layer to electrodes with a more advanced surface topology. Everywhere, the film has the same thickness and super quality.” Because of the self-limiting nature of ALD, this succeeds in a very controlled way. "Using gases and plasma, we can grow a film of one or more atomic layers in a reactor. She used a special technique, Atomic Layer Deposition (ALD), to develop film layers a few nanometers thick for this purpose. “It is important that these layers are very thin, so that they do not create extra resistance in the battery, but also uniform so that they can properly protect the electrodes,” says Hornsveld. Hornsveld therefore conducted research into making nanometer thin layers that you can apply to that interface in order to protect the electrode. This limits the performance and durability of the battery. It can become very thick and porous and consume active Li-ions. When a battery first charges, an intermediate phase is created there by the chemical reactions. The electrolyte liquid is flammable and can even explode under certain conditions."Īccording to Hornsveld, most problems arise at the interface between the electrode and the liquid electrolyte. "Li-ion batteries have a relatively high energy and power density, but given the high societal demand, that's still not high enough for many of the applications. Lithium ions can flow through the electrolyte back and forth between the two electrodes, explains PhD student Norah Hornsveld. ProblemsĬonventional rechargeable lithium-ion (Li-ion) batteries consist of two metal contacts (the plus and minus pole) with two electrodes in between and a liquid salt solution (electrolyte) in the middle. Batteries therefore play an important role in the transition to a sustainable world, which is why much research is focused on batteries that perform better, are safer, lighter, and cheaper. We also make increasing demands on our portable electrical equipment. Look at the increasing numbers of electric cars, but also e-bikes on the road: we want to drive more electrically.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
